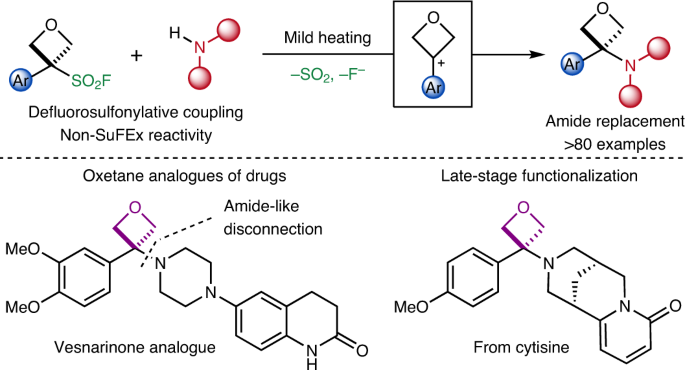
α,β‐Unsaturated Aldehydes as C‐Building Blocks in the Synthesis of Pyridines, 1,4‐Dihydropyridines and 1,2‐Dihydropyridines - Vchislo - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

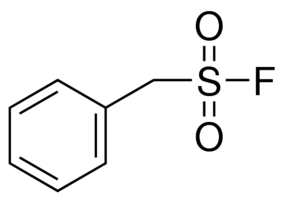
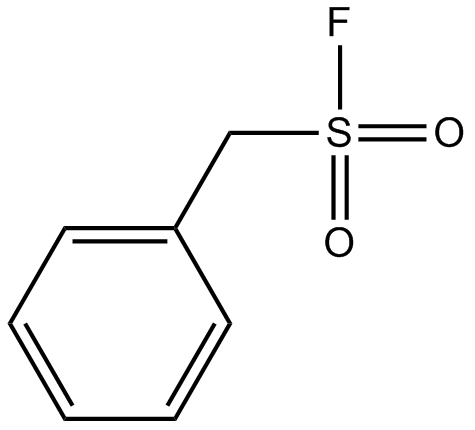
Sulfonyl fluorides as privileged warheads in chemical biology - Chemical Science (RSC Publishing) DOI:10.1039/C5SC00408J
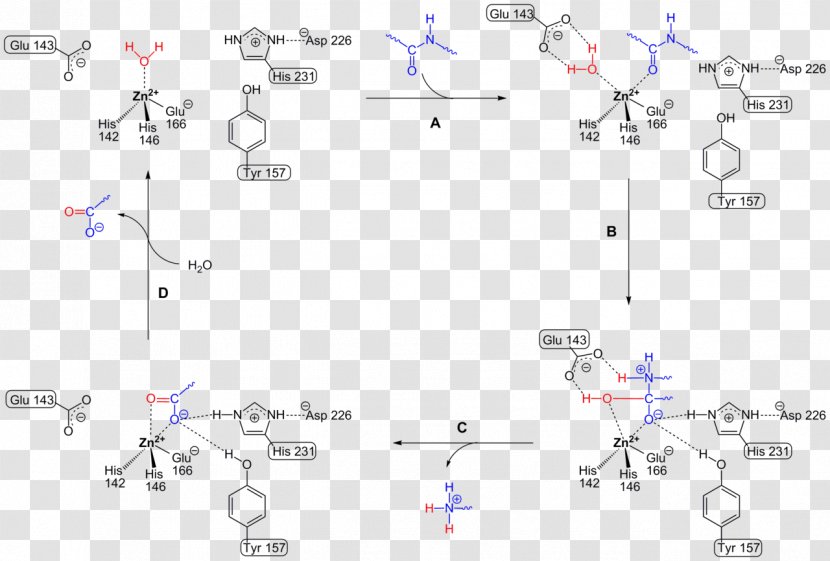
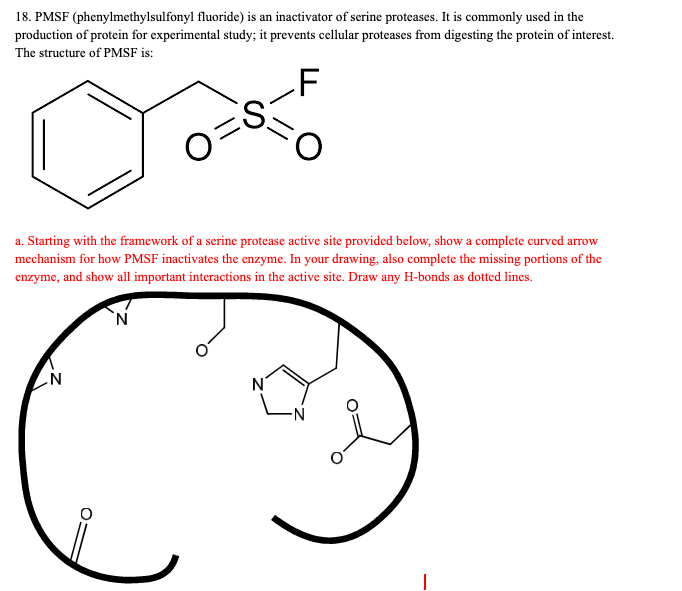
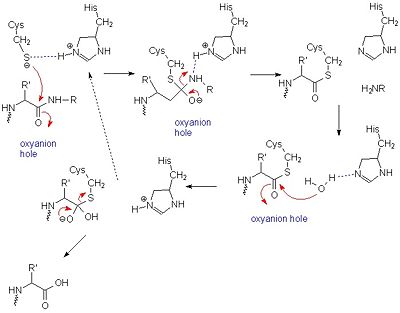
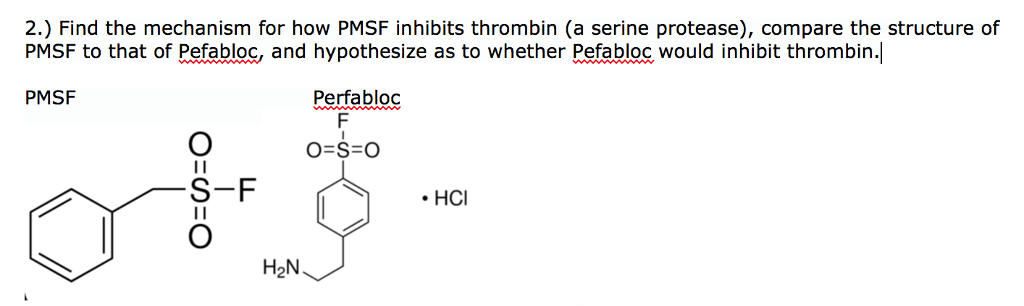
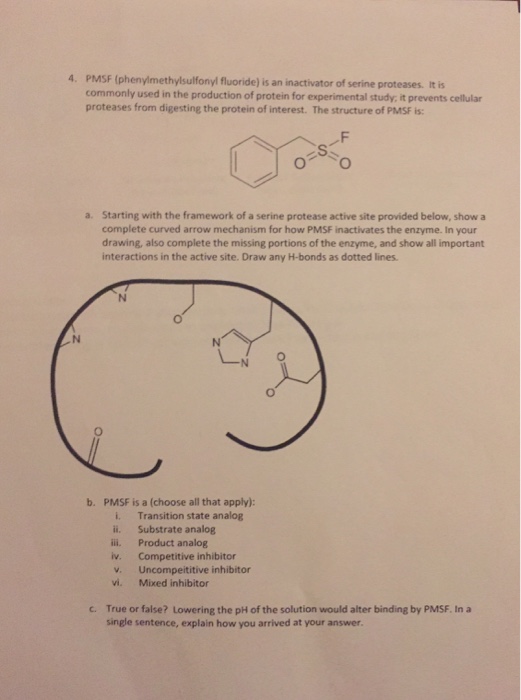
Thermolysin Metalloproteinase PMSF Protease Peptide Bond - Reaction Mechanism - Cycle Transparent PNG

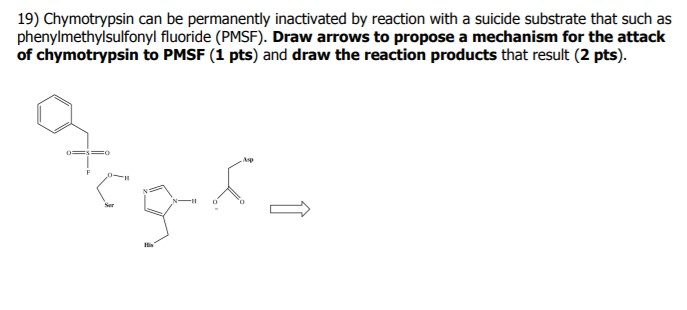
Serine protease inhibitor mediated peptide bond re-synthesis in diverse protein molecules - ScienceDirect

Figure 10 from Peptide-Based Inhibitors of Hepatitis C Virus NS3 Serine Protease: Kinetic Aspects and Inhibitor Design | Semantic Scholar
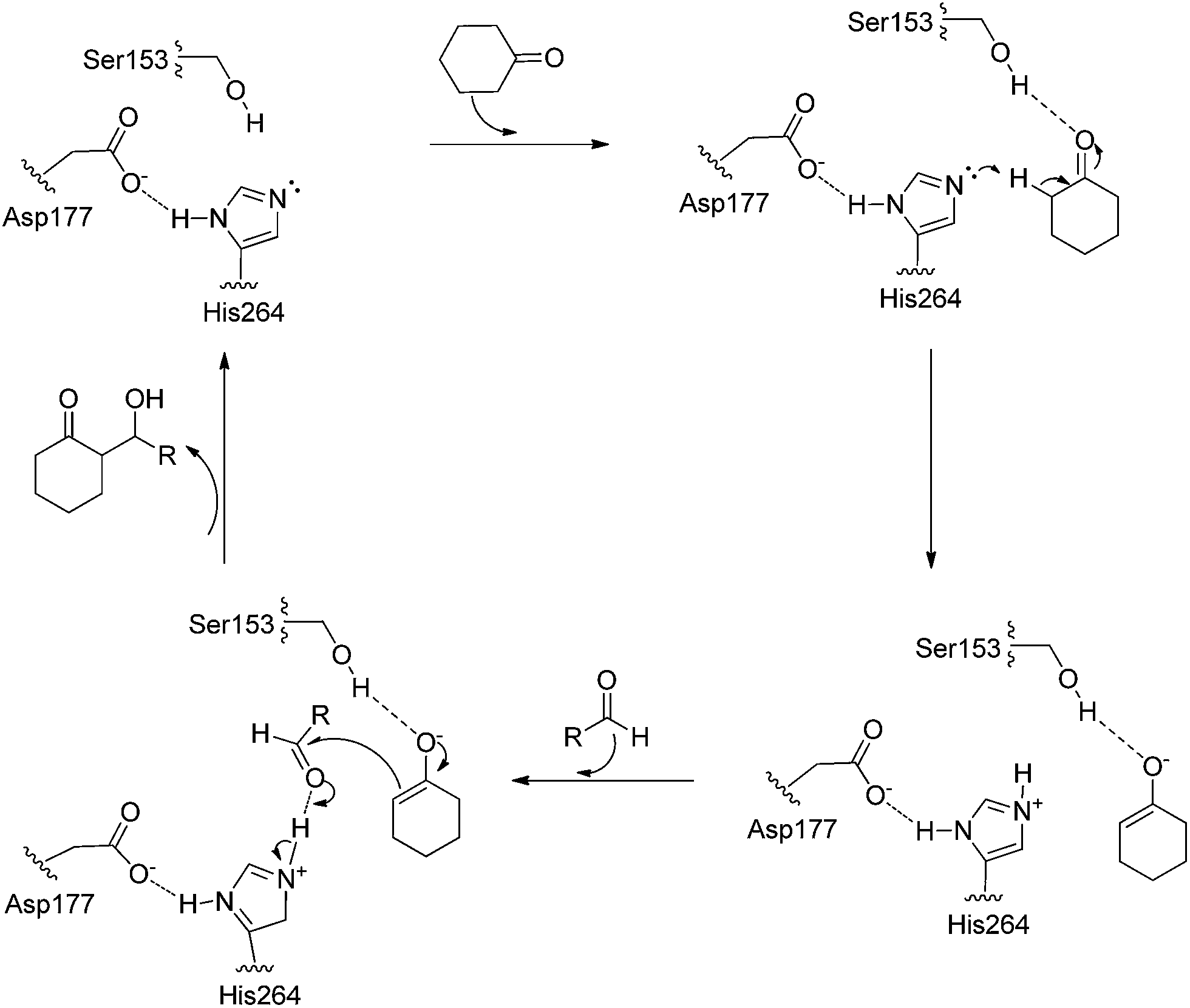
Hydrolase-catalyzed asymmetric carbon–carbon bond formation in organic synthesis - RSC Advances (RSC Publishing) DOI:10.1039/C4RA11462K
![Revisiting the Mechanism of P450 Enzymes with the Radical Clocks Norcarane and Spiro[2,5]octane | Journal of the American Chemical Society Revisiting the Mechanism of P450 Enzymes with the Radical Clocks Norcarane and Spiro[2,5]octane | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja025608h/asset/images/large/ja025608hn00001.jpeg)
Revisiting the Mechanism of P450 Enzymes with the Radical Clocks Norcarane and Spiro[2,5]octane | Journal of the American Chemical Society

Synthesis and Characterization of Diazomethylarachidonyl Ketone: An Irreversible Inhibitor of N-Arachidonylethanolamine Amidohydrolase | Journal of Pharmacology and Experimental Therapeutics
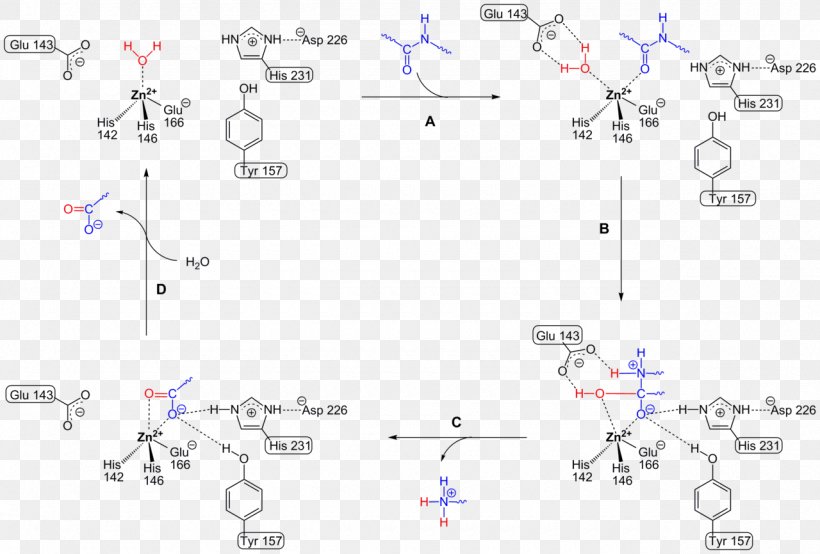
Thermolysin Metalloproteinase PMSF Protease Peptide Bond, PNG, 1280x866px, Thermolysin, Active Site, Area, Bacillus, Chemical Reaction Download
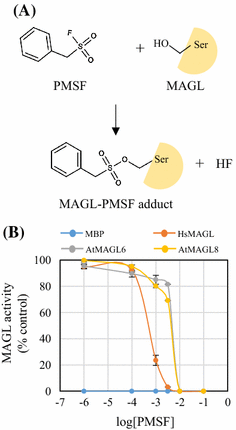
The GxSxG motif of Arabidopsis monoacylglycerol lipase (MAGL6 and MAGL8) is essential for their enzyme activities | Applied Biological Chemistry | Full Text

SciELO - Brasil - Effect of phenylmethylsulfonyl fluoride, a protease inhibitor, on enamel surface remineralization Effect of phenylmethylsulfonyl fluoride, a protease inhibitor, on enamel surface remineralization