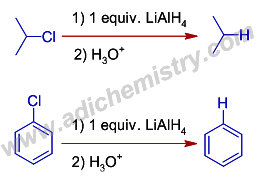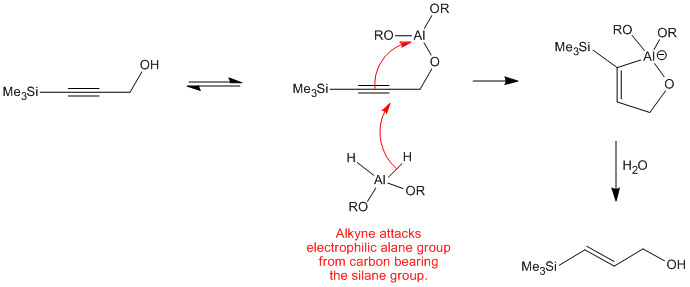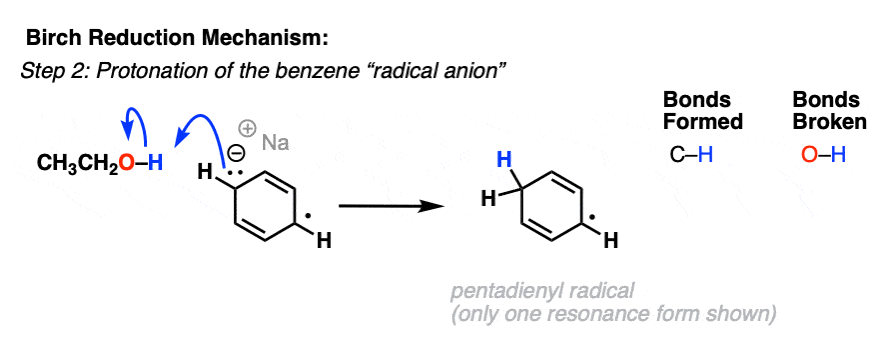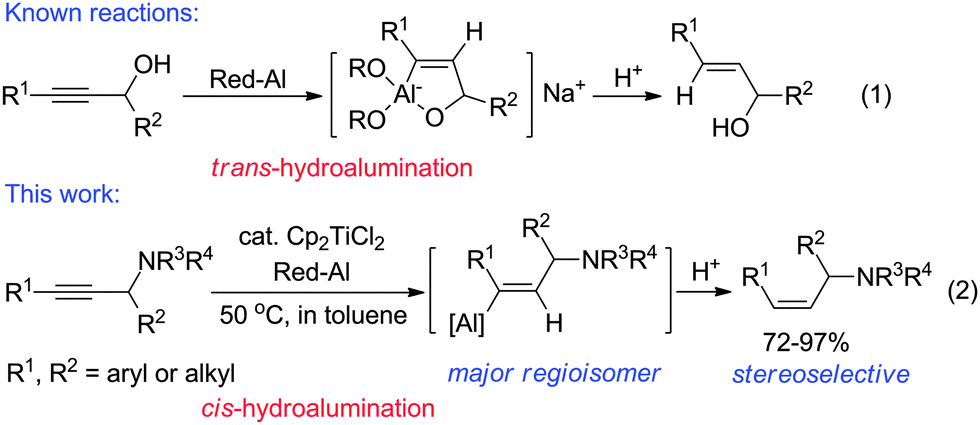
Cp 2 TiCl 2 -catalyzed cis -hydroalumination of propargylic amines with Red- Al: stereoselective synthesis of Z -configured allylic amines - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC00950B
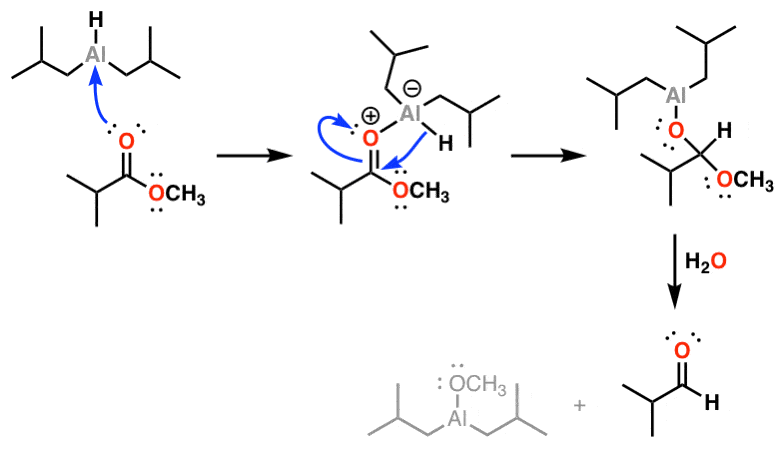
organic chemistry - Why does the reduction of a Weinreb amide give an aldehyde instead of an amine? - Chemistry Stack Exchange