
Hypervalent iodine(III): selective and efficient single-electron-transfer ( SET) oxidizing agent - ScienceDirect
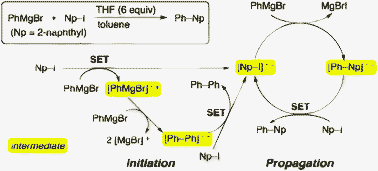
Single electron transfer-induced Grignard cross-coupling involving ion radicals as exclusive intermediates - Chemical Communications (RSC Publishing)
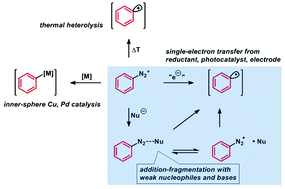
Aromatic substitutions of arenediazonium salts via metal catalysis, single electron transfer, and weak base mediation - Organic & Biomolecular Chemistry (RSC Publishing)
Scheme 3 (a) The single electron transfer mechanism for the covalent... | Download Scientific Diagram

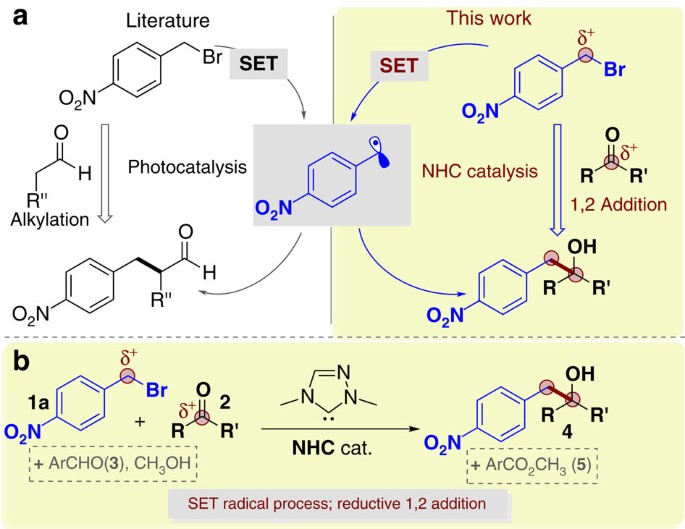
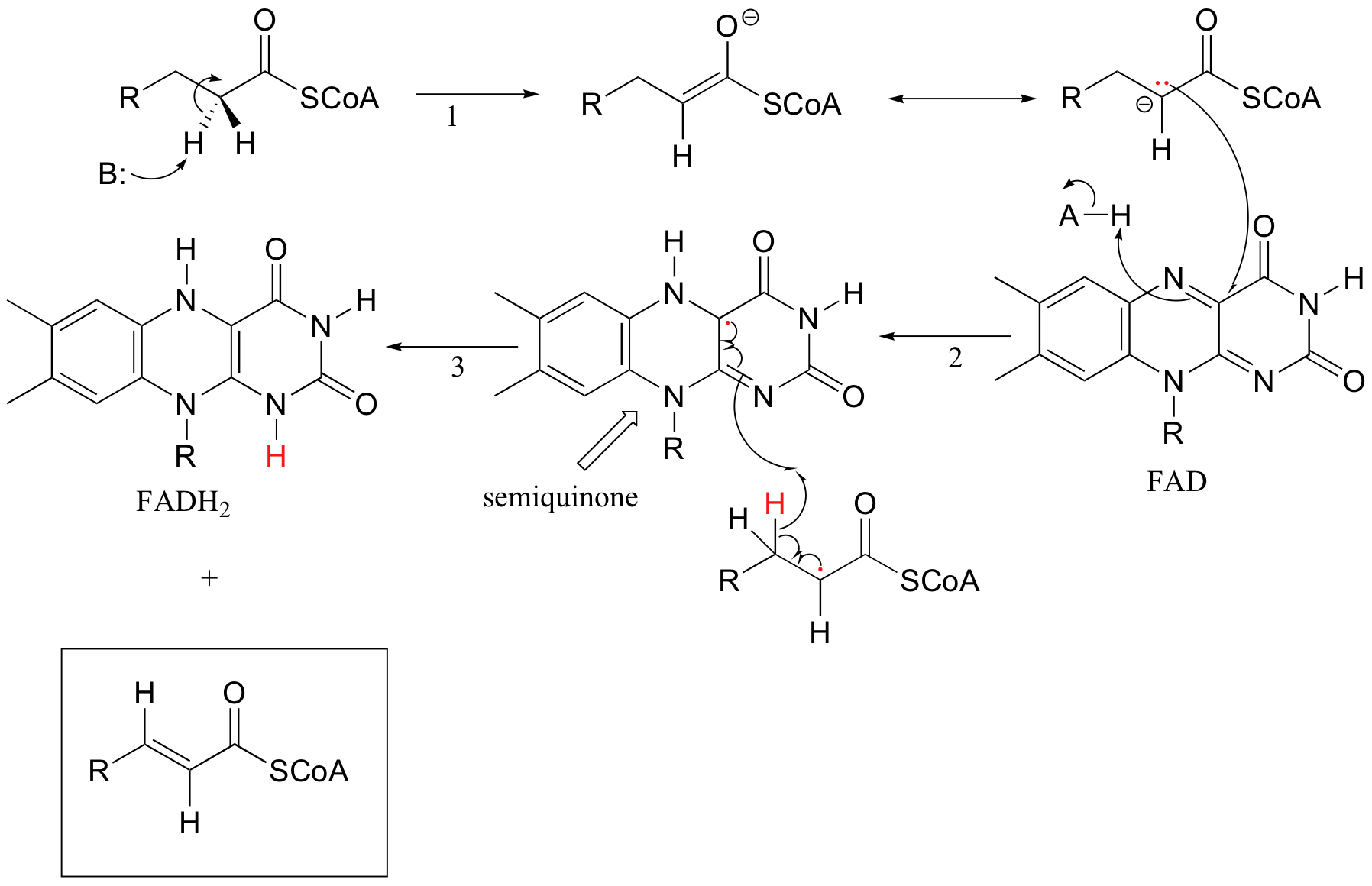
Single‐Electron Transfer Reactions Enabled by N‐Heterocyclic Carbene Organocatalysis - Li - 2021 - Chemistry – A European Journal - Wiley Online Library

Photoredox Catalysis Unlocks Single-Electron Elementary Steps in Transition Metal Catalyzed Cross-Coupling | ACS Central Science

Merging single-electron transfer and energy transfer processes of photocatalyst: An atom economical strategy for the synthesis of 1-trifluoroethylated isoquinolines from cis and trans vinyl isocyanides - ScienceDirect
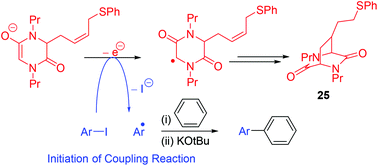
Evidence of single electron transfer from the enolate anion of an N,N′-dialkyldiketopiperazine additive in BHAS coupling reactions - Organic & Biomolecular Chemistry (RSC Publishing)

Mechanisms of antioxidant reacting with free radical: single electron... | Download Scientific Diagram

Single‐Electron Transfer Reactions Enabled by N‐Heterocyclic Carbene Organocatalysis - Li - 2021 - Chemistry – A European Journal - Wiley Online Library

Single‐Electron Transfer Reactions Enabled by N‐Heterocyclic Carbene Organocatalysis - Li - 2021 - Chemistry – A European Journal - Wiley Online Library

Single‐Electron Transfer Reactions Enabled by N‐Heterocyclic Carbene Organocatalysis - Li - 2021 - Chemistry – A European Journal - Wiley Online Library

Mechanisms of antioxidant reacting with free radical: single electron... | Download Scientific Diagram
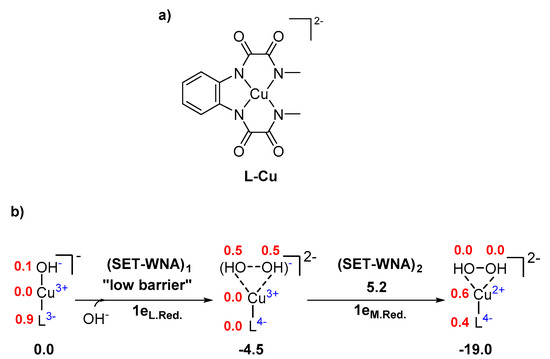
Inorganics | Free Full-Text | Computational Characterization of Single-Electron Transfer Steps in Water Oxidation | HTML



![ASAP] Mn(OAc)3-Mediated Addition Reactions of NaSO2CF3 and Perhalog ASAP] Mn(OAc)3-Mediated Addition Reactions of NaSO2CF3 and Perhalog](https://www.researcher-app.com/image/eyJ1cmkiOiJodHRwczovL3MzLWV1LXdlc3QtMS5hbWF6b25hd3MuY29tL3N0YWNrYWRlbWljL3Byb2R1Y3Rpb24vcGFwZXIvNTM5NDg3NS5wbmciLCJmb3JtYXQiOiJ3ZWJwIiwicXVhbGl0eSI6MTAwLCJ3aWR0aCI6NTEyLCJub0NhY2hlIjp0cnVlfQ==.webp)
